
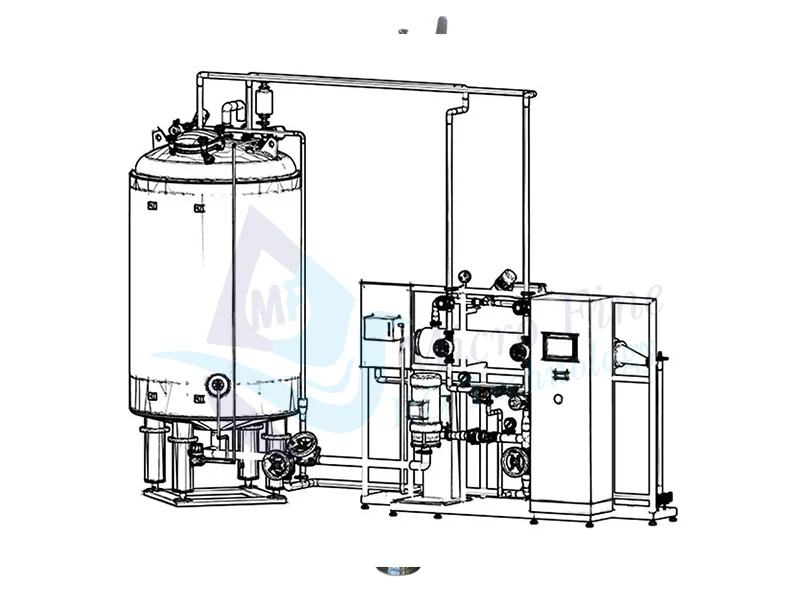
- thermal distillation
- vapor compression distillation
- combination of reverse osmosis, electrodeionization and ultrafiltration methods
Thermal Distillation:-
The process of thermal distillation is basing on evaporation and subsequent condensation of the feed water. The source of energy used to heat and evaporate the water in the process of thermal distillation can be technical steam under pressure of 4-8 bar or electric energy.
MFROT Water for Injection Plant is a fully automated, high-purity water production system designed for pharmaceutical industries. It produces water that meets USP and EP standards for sterility, endotoxin level, and conductivity. The system integrates multiple purification technologies—including reverse osmosis (RO), ultrafiltration (UF), distillation, and continuous water circulation with sanitization—to ensure water purity and regulatory compliance.
This plant is ideal for pharmaceutical manufacturing, hospitals, laboratories, and research centers where sterile, pyrogen-free water is required.
Key Features :-
- Produces USP / EP / JP compliant Water for Injection (WFI).
- Fully automated PLC control with SCADA/HMI interface.
- Multi-stage purification: RO, UF, distillation, and hot water storage.
- Continuous recirculation to maintain water quality.
- Sanitizable SS316L piping with hygienic design.
- High-quality, corrosion-resistant storage tanks.
- Real-time monitoring of conductivity, temperature, flow, and pressure.
- Optional online microbial monitoring.
- Easy operation, cleaning, and validation
Key Features :-
| Parameter | Specification (Typical) |
|---|---|
| Capacity | 500 LPH – 5000 LPH (customizable) |
| Feed Water Source | RO Water / Purified Water |
| Purification Method | RO + UF + WFI Distillation / Continuous Recirculation |
| Material of Construction | SS316L (Tanks & Piping) |
| Temperature | Hot Water Sterilization 80–90°C |
| Conductivity | < 1.3 μS/cm (as per USP) |
| Endotoxin Level | < 0.25 EU/mL |
| Automation | PLC / SCADA / HMI |
| Piping & Fittings | Tri-clamp, sanitary-grade |
| Validation & Compliance | USP / EP standards |
Why Choose Our WFI Plant?
Meets USP, EP, and WHO standards.
Top Keywords Integrated
- Water for Injection plant.
- WFI plant for pharmaceutical use.
- Sterile water production system.
- USP / EP compliant water plant.
- High-purity water system.
- Pharmaceutical water treatment plant.
Benefits
- Compliance with international pharmaceutical standards.
- Sterile, pyrogen-free water for sensitive applications.
- Continuous quality monitoring and automated operation.
- Reduces risk of contamination and ensures patient safety.
- Reliable, long-term performance with low maintenance.
- Fully automated with minimal manual intervention.
- High-quality SS316L construction for longevity.
- Easy validation, cleaning, and maintenance.
- Tailored solutions for pharmaceutical and biotech industries.
